Health Ministry Flags Faulty Cancer Drug Circulating In Kenya
On Thursday, December 5, the Ministry of Health issued a public notification recalling a cancer medicine that had previously been used for injections.
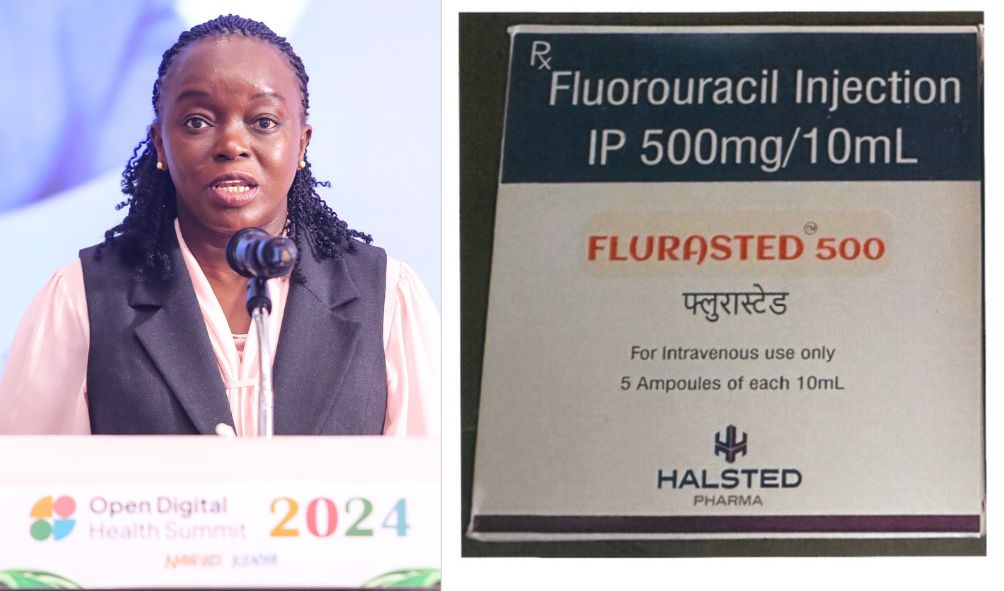
The Pharmacy and Poisons Board (PPB) issued a notice directing all health practitioners and dispensing pharmacies to immediately stop using and selling the Flurasted 500 (5-Fluorouracil) Injection.
“The Pharmacy and Poisons Board (“the Board”) orders the quarantine of Flurasted 500 (5-Fluorouracil) Injection Batch No. HHP24017 Manufactured by Halsted Pharma Private Limited, India,” the Pharmacy and Poisons Board announced in a statement.
According to the board, the decision to recall the medicine stemmed from complaints regarding its appearance, which has since caused misunderstanding.
QUARANTINE ORDER FOR SUSPECTED SUBSTANDARD FLURASTED 500 INJECTION BATCH NO. HHP24017 pic.twitter.com/c2wUdch6q7
— Pharmacy and Poisons (@ppbkenya) December 5, 2024
“The quarantine order is being issued due to a market complaint on the appearance parameter of the content. Flurasted 500 (5-Fluorouracil) Injection is used in the management of cancer,” the notice added.
Flurasted 500 (5-Fluorouracil) Injection Batch No. HHP24017 is a cancer drug used to treat several types of cancer, primarily chemotherapy.
Furthermore, the primary ingredient, 5-fluorouracil (5-FU), is a chemotherapeutic medicine that inhibits the proliferation of cancer cells and is often used to treat tumors such as colon, breast, and stomach cancer.
As a result, health practitioners have been urged to immediately discontinue the drug’s use on their patients while the board works on ways to remove it from the shelves.
“In light of this, the Board advises all pharmaceutical outlets, healthcare facilities, healthcare professionals, and members of the public to immediately quarantine the product batch and stop the further distribution, sale, issuance, or use of the affected batch,” it added.
ALSO READ:
- Orengo Responds To Claims Linda Mwananchi Fakes Teargas Chaos
- Kenya’s Omanyala Clocks Sub 10 To Win Men’s 100M In Xiamen Diamond League
- DCI, Other Security Agencies Foil Terrorist Attack & Recover IED
- EXPLAINER: Why Kenyan Shilling Could Fall To Ksh180/Dollar From Fuel Crisis
- KPA Announces New Prices For Likoni Ferry Services, Makes 50% Raise
Members of the public are also encouraged to report any suspected cases of substandard drugs to the local facility for prompt action.
The recall follows another one issued by the board on November 22, of two nasal sprays via a public alert statement.
“The Pharmacy and Poisons Board has mandated the recall of Efinox 1% w/v Batch No.82979 and Efinox 0.5% w/v Batch No. 82978 manufactured by Laboratory and Allied Ltd, Kenya,” read the statement.
The Board claimed that there was a mix-up in the labelling and packing of the two medicines, resulting in a discrepancy in their potency. As a result, the medications were judged hazardous for use.
Health Ministry Flags Faulty Cancer Drug Circulating In Kenya